We'd love to keep you informed about what is going on at Sequoia, and how our users and customers deploy and use their LISST instruments and other Sequoia products.įor that purpose, we maintain a mailing list. When ppm is used as a measure for the suspended particle concentration, it is therefore very important to specify if the concentrations are ppm BY VOLUME or ppm BY MASS, to facilitate comparisons with data where the concentrations are reported in µl/l or mg/l.ĭo you have a need for an instrument that can measure particle concentration? Have a look at our LISST-Portable|XR laser for laboratory use, our LISST-200X laser for field and submersible use, or our LISST-AOBS that measures concentration using acoustics. For example, a sample with a mass concentration of 100 mg/l will have a volume concentration of 38 µl/l. Calculate how many square centimeters are in the sheet of paper. To convert from ppm by mass to ppm by volume, divide by the density of the particles. For example, 1 ppm of a chemical in water means that, in a million units of water. If the sample is too big, the whole study becomes complex, expensive and time-consuming to run, and although the results are more accurate, the benefits don’t outweigh the costs. For example, a sample with a volume concentration of 25 µl/l will have a mass concentration of 25*2.65 = 66 mg/l. For mineral grains (clay, silt and sand sizes), this will typically be 2.65 g/cm3.

To convert from ppm by volume to ppm by mass, multiply by the density of the particles. However, if ppm is expressed as THE MASS of particles in a unit volume of water, then ppm BY MASS is equal to mg/l. If ppm is expressed as THE VOLUME of particles to a unit volume of water, then ppm BY VOLUME is equal to µl/l. For example, to convert 100 pounds per acre of phosphate fertilizer (P2O5) to ppm of elemental phosphorus, just multiply 100 by 0.5 and then by 0.4364 to.
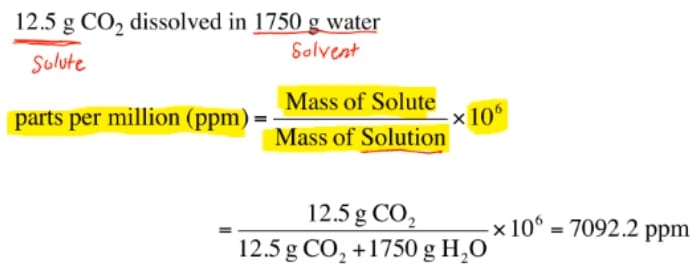
But what parts? The amount of particles in a suspension can be expressed as the total volume OR total mass of particles in a unit volume of water AND THESE TWO NUMBERS WILL ONLY BE THE SAME IF THE DENSITY OF THE PARTICLES IS 1 g/cm3.
